 Unfortunately, although he was an accomplished scientist, Avogadro was not an accomplished writer, and his hypothesis was not accepted for another 50 years. The mystery was solved in 1811 by Italian scientist Amedeo Carlo Avogadro, who argued that gaseous oxygen is composed not of atoms of oxygen but of molecules of oxygen: O 2. Many chemists, including Dalton, considered this possibility absurd: how could an atom-at the time believed to be the smallest unit of matter-split during the course of a chemical reaction? The problem with this hypothesis was that for it to be true, somehow the gaseous oxygen had to be splitting in half. The pair found numerous other simple ratios, resulting in Gay-Lussac suggesting that equal volumes of gases have equal numbers of particles, what we now refer to as Avogadro’s law. To add even more confusion, in 1805 Prussian scientist Alexander von Humboldt and French scientist Joseph Louis Gay-Lussac determined that two volumes of gaseous hydrogen always combined with one volume of gaseous oxygen to form two volumes The problem with this method is that Dalton didn’t know the numbers of oxygen atoms and hydrogen atoms in a water molecule he assumed water had a molecular formula of HO, leading to an incorrect relative atomic mass of 8 for oxygen. He then could use this ratio of 8:1 to determine the weight of oxygen compared with that of hydrogen. For example, to determine the atomic mass of oxygen, he used the fact that 1 gram of hydrogen reacts with 8 grams of oxygen to make water. So how did chemists of the 19th century define atomic mass? In 1803, English scientist John Dalton published an article in which he assigned hydrogen a weight of 1, and then used compounds of hydrogen to determine the relative weights of the other elements. The idea of isotopes wasn’t introduced until 1913, and neutrons weren’t discovered until 1932. It would be 30 years after Mendeleev’s periodic table that scientists found out atoms were composed of smaller bits and pieces. In fact, many chemists held the view that atoms were the smallest units of matter possible. The modern definition of atomic mass (the weighted average of the atomic masses of all isotopes of an element) was meaningless 150 years ago. Here is where we will begin our journey to periodicity. One such discovery was that of atomic masses. The modern periodic table didn’t spring fully formed from the genius of Mendeleev it was shaped by key discoveries about the elements. So how did Russian chemist Dmitri Ivanovich Mendeleev and the other creators of the periodic table (arguably six of them) bring order to the elements? How did they create a tool that would ultimately house 118 elements when they knew only 62 of them? And why does Mendeleev get all the credit? Mass + reactions = periodic table It even predates knowledge of the noble gases. 
However, the periodic table predates knowledge of atomic numbers and subatomic particles (yes, including electrons). If you know about atomic numbers and electron shells, recreating the periodic table is simple. 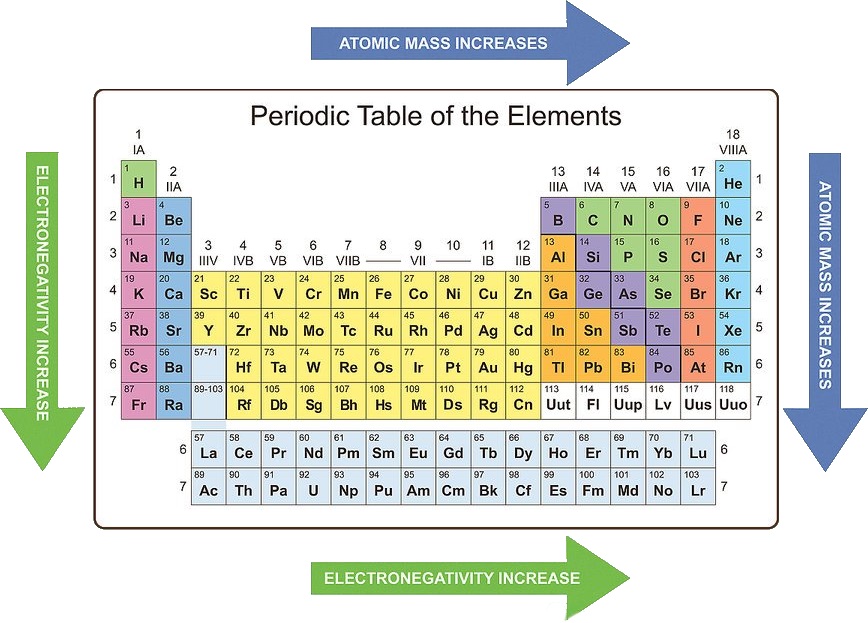
You would order them by increasing atomic number and create a new row when you hit a noble gas. If I gave you all of the elements on cards and told you to recreate the periodic table, you probably wouldn’t have much trouble. You can compare electronegativity, ionization energy, atomic radius, chemical reactivity, and more. 
You can determine the electron configuration of any atom, simply from its place. 
The periodic table is an elegant demonstration of properties of elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
